CAP-Gly domain
From Proteopedia
| |||||||||
| 1tov, resolution 1.77Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | |||||||||
| Gene: | F53F4.3 (Caenorhabditis elegans) | ||||||||
| |||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum, TOPSAN | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Contents |
CAP-Gly domains in cytoskeletal proteins
Activity
The cytoskeletal network contains Cytoskeleton-Associated Proteins (CAPs), including CLIP-170 and dynactins, that function in the organization of microtubules and in the transportation of vesicles and organelles. CAPs that bind to the +end of microtubules generally contain one or more glycine-rich domains ("CAP-Gly" domains) with a well-conserved GKNDG sequence motif[1], presumably involved in protein-protein binding[2].
Structure
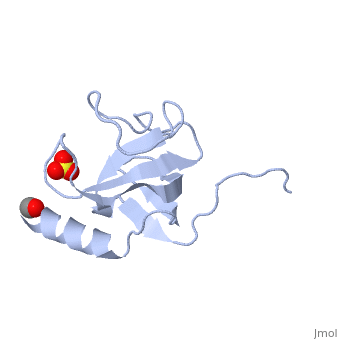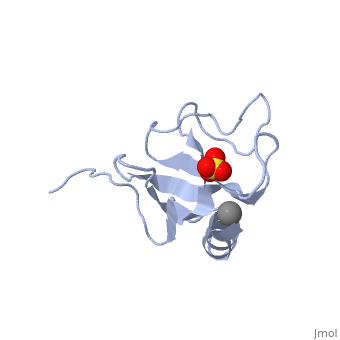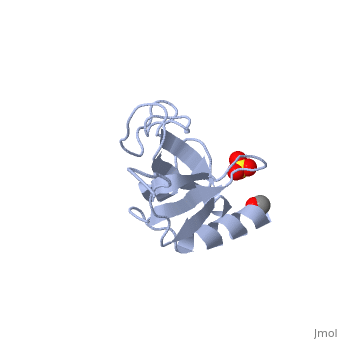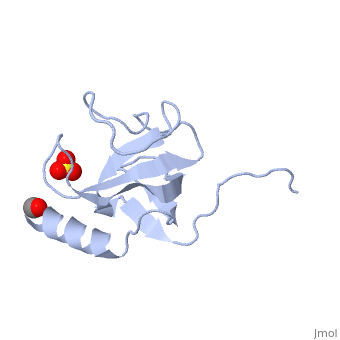
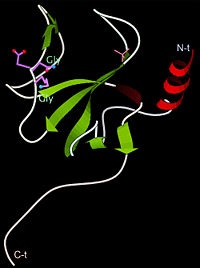
The first crystal structure of a CAP-Gly domain (PDP file 1lpl), from C. elegans protein F53F4.3, revealed a novel protein fold containing a small 5-strand antiparallel beta-barrel, several loops, and an N-terminal helix. The GKNDG sequence is in two consecutive turns of a surface loop, at one side of a groove (as shown in image at left). The groove is lined by a large, concave patch of residues highly conserved among the related sequences[3]. In the crystal, a dimer is formed by binding of the C-terminus of each chain into the GKNDG groove of the other chain.
1TOV is a rebuilt and re-refined version of the same dataset from 1LPL, giving an improvement of about 4% in Rfree, identifying a sulfate near the groove (seen in both 2D and interactive images), and adding 3 more residues of ordered helix toward the N-terminal end of the domain fragment. Both are from the SouthEast Collaboratory for Structural Genomics (SECSG), solved by single wavelength sulfur-anomalous phasing. The new 1tov structure was produced as part of a systematic 30-structure test of the then-new MolProbity methods for diagnosing and correcting problems in crystallographic models[4].
As of 2012 there were 23 CAP-Gly domains in the PDB, including CLIP-170 domain 2 (2e3h) at 1.45A resolution.
Crystal structures
- 2e3h, 2e3i CLIP-170, or restin
- 1tov, 1lpl C. elegans hypothetical
- 1txq p150Glued
- 2hqh dynactin/restin
Solution NMR structures
- 1ixd, 1whl, 1whm domains in human CYLD
- 1whg tubulin-specific chaperone
- 1whh, 2cp0 CLIPR-59
- 1whj, 1whk domains 1, 3 mouse hypothetical
- 2cow KIF-13B
- 2coy dynactin-1
- 2coz CAP350
- 2cp2, 2cp3 CLIP-115
- 2cp5, 2cp6, 2cp7 restin
- 2e4h restin/tubulin
References
- ↑ Riehemann K, Sorg C. Sequence homologies between four cytoskeleton-associated proteins. Trends Biochem Sci. 1993 Mar;18(3):82-3. PMID:8480366
- ↑ Pierre P, Scheel J, Rickard JE, Kreis TE. CLIP-170 links endocytic vesicles to microtubules. Cell. 1992 Sep 18;70(6):887-900. PMID:1356075
- ↑ Li S, Finley J, Liu ZJ, Qiu SH, Chen H, Luan CH, Carson M, Tsao J, Johnson D, Lin G, Zhao J, Thomas W, Nagy LA, Sha B, DeLucas LJ, Wang BC, Luo M. Crystal structure of the cytoskeleton-associated protein glycine-rich (CAP-Gly) domain. J Biol Chem. 2002 Dec 13;277(50):48596-601. Epub 2002 Sep 7. PMID:12221106 doi:http://dx.doi.org/10.1074/jbc.M208512200
- ↑ Arendall WB 3rd, Tempel W, Richardson JS, Zhou W, Wang S, Davis IW, Liu ZJ, Rose JP, Carson WM, Luo M, Richardson DC, Wang BC. A test of enhancing model accuracy in high-throughput crystallography. J Struct Funct Genomics. 2005;6(1):1-11. PMID:15965733 doi:10.1007/s10969-005-3138-4