User:Karl E. Zahn/RB69 DNA polymerase (GP43)
From Proteopedia
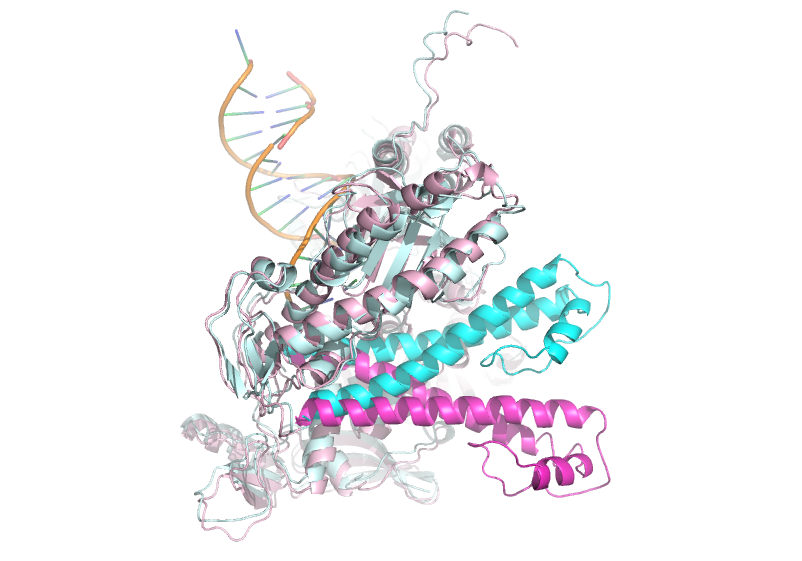
DNA polymerases (pols) are a specialized class of transferase enzymes that catalyze template directed extension of a primed DNA strand from free deoxynucleoside triphosphates. The family B DNA pol encoded in the genome of the bacteriophage RB69 (gp43) represents such an enzyme that shares significant homology with the human primase associated pol α, the human replicative pols δ and ε, and the human repair pol ζ. The relative ease with which one can induce RB69 pol to form diffraction quality crystals in complex with DNA, in addition to its homology shared with important human enzymes, has allowed RB69 pol to serve as an extremely useful structural tool and model enzyme.
|
The first crystal structure of RB69 pol, solved in 1997, revealed the domain topology characteristic of polymerases: the palm, fingers, and thumb domains obtain a spatial arrangement reminiscent of a . RB69 pol also wields intrinsic 3'→5' exonuclease proofreading activity, which drastically improves the fidelity of this enzyme and occurs in most replicative pols. An N-terminal domain precedes the exonuclease domain in primary sequence, bringing the total to .[1]
Contents |
Fingers
The of RB69 DNA pol exist as an insertion between the two sequences forming the palm. Since the fingers domain experiences a 60 degree rotation from the open or apo structure to the closed catalytically competent structure, the fact that the palm flanks the fingers in primary sequence is likely required for proper communication between these domains. The fingers domain also provides important (K560, K486, R482) that bind triphosphates. K560 is also believed to stabilize the pentacovalent intermediate formed with nucleophilic attack of the 3'-OH on the alpha phosphate[2].
An additional role of the fingers domain, which has not received thorough investigation at this point, is the manner in which this domain in stabilizes the template DNA strand. The residue provides stacking interactions upstream of the templating position in several structures. In addition to other aromatic residues spatially adjacent to this position, interactions to W574 could be important for catalysis in some situations.
Palm
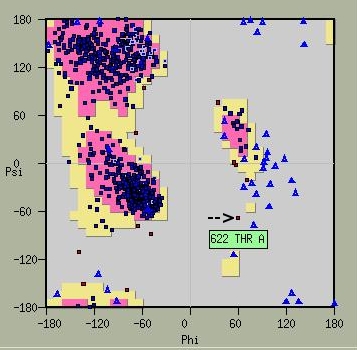
The two-metal ion mechanism employed during the DNA pol catalytic cycle relies on coordination of divalent cations by conserved aspartate motifs in the [3][4]. The palm domain of RB69 DNA pol (residues 383-468, 573-729) contains such (LDFASLYPSI and DTDS) that are particular to family B DNA pols. Interestingly, the residue T622 in this sequence must obtain a high energy conformation in order to correctly orient the D621 and D623. This property is evident in Ramachandran plots of RB69 DNA pol where T622 is an obvious out-layer. L411 and D412 complete the coordination sphere of metal ion B.
A common strategy for crystallizing DNA polymerases in complex with both DNA and an incoming nucleoside triphosphate utilizes a dideoxy-terminated primer strand in the DNA molecule. Since the 3'-OH is absent, the reaction can not proceed following binding and alignment of the substrates. However, since the 3'-OH is required to complete contacts to the coordination sphere of metal ion A, complexes with chain terminated primers often illustrate perturbation or absence of this ion[5]. Metal B is absent in the structure 2OZS.
An additional important residue of the palm domain is . Since polymerases monitor the duplex DNA in a sequence independent manner, contacts to the minor groove are required for catalysis and translocation of the templating strand. K706 protrudes into the minor groove of the DNA and provides direct contacts from the protein to the DNA. Important water-mediated contacts also serve this purpose. Image:GP43 Minor-Groove.png
Exonuclease domain
Intrinsic exonuclease proofreading activity elevates the fidelity of DNA synthesis. While an exonuclease deficient mutant is generally required for co-crystallization experiments, the DNA is still resolved in the active site in some situations. This is illustrated when a lesion of base loss resides in the pol active site, where half of the molecules of the asymmetric unit often obtain a proof-reading conformation (pdbid 2OYQ, 2P5G, or 2PO5 for examples)[6][7].
References
- ↑ Wang J, Sattar AK, Wang CC, Karam JD, Konigsberg WH, Steitz TA. Crystal structure of a pol alpha family replication DNA polymerase from bacteriophage RB69. Cell. 1997 Jun 27;89(7):1087-99. PMID:9215631
- ↑ Franklin MC, Wang J, Steitz TA. Structure of the replicating complex of a pol alpha family DNA polymerase. Cell. 2001 Jun 1;105(5):657-67. PMID:11389835
- ↑ Steitz TA, Steitz JA. A general two-metal-ion mechanism for catalytic RNA. Proc Natl Acad Sci U S A. 1993 Jul 15;90(14):6498-502. PMID:8341661
- ↑ Doublie S, Tabor S, Long AM, Richardson CC, Ellenberger T. Crystal structure of a bacteriophage T7 DNA replication complex at 2.2 A resolution. Nature. 1998 Jan 15;391(6664):251-8. PMID:9440688 doi:http://dx.doi.org/10.1038/34593
- ↑ Batra VK, Beard WA, Shock DD, Krahn JM, Pedersen LC, Wilson SH. Magnesium-induced assembly of a complete DNA polymerase catalytic complex. Structure. 2006 Apr;14(4):757-66. PMID:16615916 doi:http://dx.doi.org/10.1016/j.str.2006.01.011
- ↑ Zahn KE, Belrhali H, Wallace SS, Doublie S. Caught bending the A-rule: crystal structures of translesion DNA synthesis with a non-natural nucleotide. Biochemistry. 2007 Sep 18;46(37):10551-61. Epub 2007 Aug 24. PMID:17718515 doi:10.1021/bi7008807
- ↑ Hogg M, Wallace SS, Doublie S. Crystallographic snapshots of a replicative DNA polymerase encountering an abasic site. EMBO J. 2004 Apr 7;23(7):1483-93. Epub 2004 Apr 1. PMID:15057283 doi:http://dx.doi.org/10.1038/sj.emboj.7600150