User:Tilman Schirmer/Sandbox 201
From Proteopedia
PleD
Contents |
Overview
|
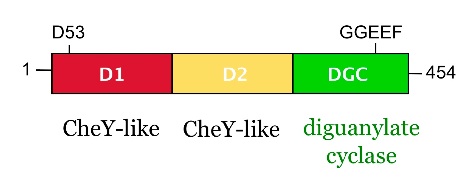
from Caulobacter crescentus is a response regulator with an unorthodox catalytic, diguanylate cyclase, output domain. It is composed of a canonical CheY-like response regulator receiver () domain,
a Rec-like () adaptor domain,
and a C-terminal domain that confers the catalytic acitvity.
The GGDEF domain is named after the highly conserved (in PleD it is GGEEF) that locates to a β-hairpin.
Substrate binding
|
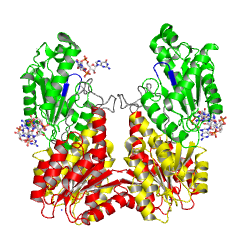
The motif is part of the as identified in the structure of PleD in complex with . The GGDEFY domain binds only one GTP subsrate molecule. For the reaction to proceed, two GTP loaded GGDEF domains have to align antiparallely. MODEL.
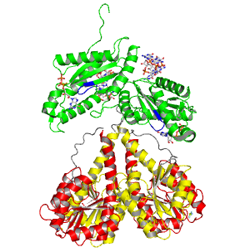
Allosteric product binding site
|
C-di-GMP
Primary inhibition site (Ip)
Secondary inhibition site (Is)
Primary and secondary inhibition sites
Two conformations
|
|