Introduction
The human glucagon receptor (GCGR) is one of 15 secretin-like, or Class B, G-protein coupled receptors (GPCRs). Like other GPCRs, it has a helical domain and a globular N-terminus (ECD).
Function
The glucagon receptor plays an important role in glucose homeostasis. During times of fasting (or low blood sugar) the pancreas produces glucagon to activate the GCGR in the liver. The binding of glucagon to the extracellular side of GCGR leads to the activation of the receptor. On the intracellular side, upon this activation, a guanine diphosphate (GDP) is exchanged for a guanine triphosphate (GTP) - which, in turn, activates adenylate cyclase. Converting adenosine triphosphate (ATP) into cyclic adenosine monophosphate (cAMP), adenylate cyclase initiates protein kinase A (PKA) activity, which releases glucose into the blood stream. Overall, activation of the GCGR elevates blood sugar levels.
Structure
Class B vs. Class A
Like all classes of glucagon receptors, which include class A (rhodopsin-like), B (secretin-like), and C (metabotropic glutamate), GCGR has a 7tm domain. While class B receptors do share characteristics with class C receptors, they are more similar to class A receptors. Class B receptors and class A receptors share less than 15% sequence homology; however, they do share similar signal transduction mechanisms as well as the 7tm domain. The orientations and positions of the 7tm helices are also conserved between both classes of glucagon receptors[1].
However, one particular difference between class A receptors and class B receptors is an inward shift of the intracellular component of Helix VII. In class A receptors this inward shift is instrumental in receptor activation, yet in class B receptors it remains unclear what role this shift plays[1].
In contrast to class A glucagon receptors which have a proline kink, in all secretin-like class B glucagon receptors a Glycine at position 393 in Helix VII allows for a . This glycine helical bend is fully conserved in all secretin-like class B receptors.
Another important structural component found in all secretin-like class B receptors are the two conserved salt bridges found between Arg 346 and Glu 406 and Arg 173 and Glu 406. These are a distinct feature of class B receptors because their interaction results in the distinct stalk found only in class B receptors[1].
While interface interactions between helices VI, V, and III, are not unique to class B receptors because certain homologous and even conserved residues exist in class A receptors (like Tyr 239 and Leu 358), as a part of the interface between helices VI, V, and III, a Class B-specific hydrogen bond occurs between Asn 318 of Helix V and Leu 242 of Helix III [1].
GCGR-Specific Traits
Helix I Stalk Region
The tip of Helix I extends above the cell membrane into the extracellular space creating a . This region is longer than any other class of GPCR and extends three α-helical turns above the plane of the membrane. It is proposed that the stalk helps to capture the glucagon peptide and facilitates it's insertion into the 7tm[1].
Intracellular Helix VIII
The GCGR also contains an intracellular Helix VIII that is comprised of roughly 20 amino acids at the C-terminal end. This helix tilts approximately 25 degrees away from the membrane - the corresponding position in class A receptors are turned toward the membrane[1]. Although researchers are not entirely sure of its function, this helix is completely conserved in class B structures.
Binding Pocket
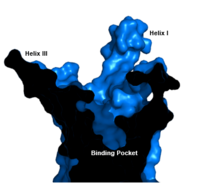
Figure 1. GCGR Binding Pocket. A cross-section of the GCGR binding pocket shows its width and depth
The class B GPCR has the widest and longest of all other classes of GPCRs. The distance between the EC tips of Helicies II and VI as well as between the tips of Helicies III and VII are some of the largest among the GPCRs
[1]. As a result, the
binding cavity of GCGR is located deeper inside the receptor, meaning glucagon binds much closer to the cell membrane.
Other Unique Structural Features
An important interface stabilization interaction between Helices I and VII occurs between Ser 152 of Helix I and Ser 390 of Helix VII. Due to their close proximity to one another, they form an important which stabilizes the structure of GCGR. Mutations to the homologous residues Ser 135 and Ser 392 have been shown to alter receptor signaling in glucagon-like peptide-1 receptor (GLP1R).
Glucagon Binding
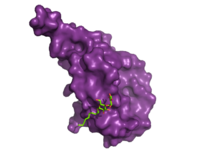
Figure 2. Bound Molecule of Glucagon. A molecule of glucagon is shown bound to the GCGR's ECD (shown in magenta)
Research has shown that class B GCPRs exist in either an
open or closed conformation differentiating between the receptor's active and inactive states. The active, or open conformation, is characterized by an intracellular outward movement of (breaking hydrogen bonds between and )
[2] and an extracellular rotation of the ECD until it is almost perpendicular to the membrane surface
[3]. While the stalk region of Helix I helps to facilitate the motion of the ECD, intracellular G-protein coupling and extracellular glucagon binding stabilized this active state. In the abscence of glucagon, however, the GCGR adopts a closed conformation in which all three of the extracellular loops of the 7tm () can interact with the ECD
[3]. In this closed state, the ECD covers the extracellular surface of the 7tm. To transition between states, the ECD rotates and moves down towards the 7tm domain. This transition mechanism is consistent with the "two-domain" binding mechanism of class B GCPRs in which (1) the C-terminus of the ligand first binds to the ECD allowing (2) the N-terminus of the ligand to interact with the 7tm and activate the protein
[2].
Clinical Relevance
Because of GCGR's role in glucose homeostasis, GCGRis a potential drug target for Type 2 diabetes. Specifically, molecules that antagonize the glucagon receptor may be able to lower blood sugar levels. Among experimental treatments, two antibodies, mAb1 and mAb23, target the ECD domain of the GCGR interrupting glucagon binding[4]. While the entire cleft of the ECD is blocked by mAb1, mAb3 blocks glucagon binding by stabilizing a conformation of the ECD that promotes receptor inactivation [4]. Another antibody, mAb7, inhibits GCGR allosterically[5]. Through binding to a site outside of the binding pocket, mAb7 inhibits the receptor without interacting with essential glucagon binding residues. Disrupting the normal interactions between the ECD and the 7tm domains, these antibodies inhibit the receptor's function and help to lower blood glucose level.
As GCGR holds great promise as a therapeutic target, there are currently three drugs under development that are designed to treat Type 2 Diabetes by targeting the human glucagon receptor[2]. Although they are still in phase I of their clinical trials, preliminary research has shown promising results[6].


