Introduction
Histones
Histones are a family of basic, positively charged proteins that associate with DNA inside the nucleus to help condense the DNA into chromatin [1]. The nuclear DNA is wrapped around the histone in order to fit in the nucleus. Nucleosomes are chromatin beads made up of DNA wrapped around eight histone proteins, or a histone octamer [1]. Four different examples of modifying histones including Histone acetylation, Histone deacetylation, Histone methylation and Histone demethylation [1].
Histone Deacetylases (HDACs)
ε-Amino-lysine acetylation is a type of histone modification that controls the stability of proteins and biological function in eukaryotic cells [2]. Histone Deacetylation is the reversal process for this acetylation modification. There are different classes of HDACs based on phylogenetic analysis:
•Class I - HDACs 1-3 and 8, which are homologous to yeast Rpd3
•Class II - HDACs 4-7, 9 and 10, which are homologous to yeast Hda1
•Class III - Sirtuin deacetylases
•Class IV - HDAC 11 [2].
HDACs 1-11 are metalloenzymes and require a zinc ion for deacetylation [2].
HDAC8
Histone Deacetylase 8 is
Structure
General Structure Information
Inhibitor
Potassium Binding Site
Deacetylation
Zn2+ Metal Ion Mechanism
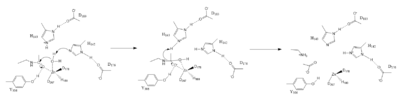
Figure 1. Mechanism of HDAC8
Active Site
Disease
HDACis

