User:Charli Barbet/Sandbox
From Proteopedia
Grb2 (1gri)
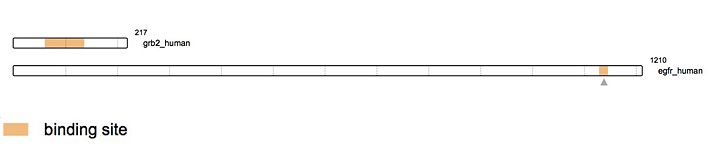
Growth Factor Receptor Bound Protein (Grb2) is a cytosolic protein made of 217 amino acids and weighing 25,206 Da. Ubiquitously present in the cell, the protein is involved in signal transduction and plays a major role in the MAP kinase pathway. Grb2 interacts mainly with tyrosine kinases such as EGFR. When EGFR is activated by ligand binding, a Guanine Nucleotide Exchange Factor GEF is recruited (like SOS1), stimulating the activation of other pathways. Several others interactions have been elucidated like the capacity of the protein to dimerise proving its potential implication in the growth of malignant cells.
StructureGrb2 protein has a very well characterized structure. Composed of 217 amino acids organized in two chains structured in β sheets and α helices. The protein has three main domains: -
-
- Another 60 amino acids long
: SH2 domain is a domain that is approximately 100 amino acids long with a very conserved structure. It has been identified in several human and rodent proteins such as phosphatases, transcription factor, or adaptor protein like Grb2. This domain is ubiquitous in several protein implicated in cellular signaling pathways.Typically, the SH2 domain specifically recognizes sites with phosphorylated tyrosines. SH2 can, for instance bind to the intracellular region of EGF leading in turn, to the formation of protein signalization complexes. This binding and the role of SH2 is very important in the conversion of an extra-cellular signal in an intra-cellular signal giving rise to diversified cellular responses or the expression of specific genes. It is also important to note that the SH2 domain can bind to other SH2 domains. Nevertheless, a mutation in the specific binding site of SH2 can impede the interaction of two proteins and thus the formation of a protein complex. Therefore, mutations in SH2 can give rise to cellular dysfunction and lead to several diseases. [1] : The SH3 domain is approximately 50 amino acid long. Largely expressed in proteins associated with the membrane. The domain is made of 5 to 6 β-sheets arranged in two antiparallel β-sheets. The linking region between the two β-sheets is made of α helices. This special conformation allows the arrangement of a hydrophobic pocket in which the ligand can bind. Typically, the binding region has a motif rich in Prolines: PXXP. This binding allows the formation of multi-protein complexes involved in the translation and conversion of extra-cellular signals. The binding is thus largely involved in gene expression and protein concentration. [2] ISOFORM: Grb2 posses an isoform, known as Grb3.3. Grb3.3 is present in cells but it induces apoptosis. The isoform has a very similar structure to Grb2 but is truncated from the 60th to the 100th amino acid resulting in a degradation of the SH2 domain and a loss of functionality. [3] FunctionThe Grb3.3 isoform has a non-functional SH2 domain, unable to bind the phosphorylated tyrosines of its targeted protein (EGFR for instance). The inability of the molecule to transmit signal is translated by apoptosis of the cell, thus regulating growth signal. The functional isoform: Grb2, is involved in several cellular functions detailed below: On one hand, the SH2 domain recognizes phosphorylated residues which are mainly tyrosines. The recognized tyrosines present a caracteristic motif for recognition: NH2-pYXNX-COOH. - pY representing the phosphorylated tyrosine. - N for Asparagine - X for a random residue By the special recognition of this motif, the binding of the two molecules is very specific. These motifs are highly expressed in several cellular proteins like Receptor Tyrosine Kinase (epidermal growth factor receptor, fibroblast growth factor receptor) but equally in proteins that are not Receptor Tyrosine Kinase (focal adhesion kinase, insulin receptor substrate-1). The recognition of phosphorylated tyrosine on the intracellular domain of Growth Factor Receptors by SH2 domain of Grb2 can lead to the recruitment of SOS-1on the SH3 side of Grb2 able to recognize the proline rich region of SOS-1 protein (Son Of Sevenless). Following this pathway and the formation of a complex between Grb2 and SOS, the RAS protein is activated. Interestingly, RAS is a G-protein implicated in the activation of RAF-1. The latest activates the MEK downstream cascade pathway (MEK1/ MEK2 and ERK1 / ERK2) involved in the translocation of ERK factors from the cytosol to the nucleus for the activation of Elk-1 and Myc transcription Factor. These particular transcription factors participate in the activation of SRE containing gene leading to cellular growth. [4] On the other hand, in T lymphocytes, the simulation of TCRs induces tyrosine phosphorylation on a wide range of cellular proteins such as p36-p38 or LAT. As an example, the phosphorylated residues of LAT can bind the SH2 domain of Grb2 while the formation of this complex recruits on the SH3 domains of Grb2 some proteins of the VAV family. VAV proteins are guanine nucleotide exchange factors (GEF) for the GTPase proteins of the Rho family. The formation of this specific complex introduces a Calcium flux and activates MAP kinase allowing T lymphocyte proliferation.[5] Finally, it was proven that Grb2 plays a role in the negative regulation of EGFR. Indeed, c-Cbl is a protein implicated in the E3 complex of EGFR ubiquitination.C-Cbl thanks to its SH2 domain can directly bind to EGFR causing its degradation (Grb2 independent regulation). Or, c-Cbl can also indirectly bind to EGFR via its SH3 domain recognition by Grb2 (dependant Grb2 regulation). The direct or indirect binding of c-Cbl on EGFR induces the recruitment of enzymes that are necessary for the ubiquitination of EGFR. Ubiquitination being a signal for protein degradation. It is important to note that negative regulation is more important when Grb2 is implicated and bound to c-Cbl rather than when c-Cbl is the only protein involved. [6] [7]
InteractionsSos1: Promotes the exchange of Ras-bound GDP into GTP, by promoting the Ras specific guanine nucleotide exchange factor activity. [8] Shc: Shc is important in the regulation of apoptosis and drug resistance in mammalian cells. It is implicated in the EGFR pathway. [9] Cbl: Cbl is a proto oncogene protein which serves as an adaptor and a negative regulator of many signalling pathways implicated in cell surface receptors activation. [10] Gab2: Gab2 acts downstream of several cell surface receptors such as cytokine, hormone, cell matrix or growth factor receptor. Thus, it is implicated in many different pathways. [11] LCP2: Involved in T cell antigen receptor mediated signaling. [12] Erbb2: Erbb2 is a kinase involved in several surface receptor complexes, but need a co-receptor for ligand binding. For example, it participates in neuregulin receptor complex but it can’t bind to it on its own. [13] Frs2: Fibroblast growth factor receptor substrate 2 can bind to FGR and NGF activated receptor. They play an important role in the activation of MAPK kinase, or the phosphorylation of PIK3R1. [14] Irs1: [Insulin receptor substrate 1 may mediate the control of various cellular processes by insulin. It can activate the phosphatidylinositol 3 kinase when it binds to the regulatory p85 subunit. [15] Gab1: GRB2 associated binding protein 1, is implicated in many signalling cascades triggered by activated receptor type kinases. It is also probably involved in signalling by the epidermal growth factor receptor. [16] EGFR: The epidermal growth factor receptor has a Tyrosine kinase activity and can be recognized by Grb2 thanks to its Tyrosine domains. This receptor is implicated in many pathways, such as antigen fixation on B cells. [17]
EGFR interactionAs stated earlier, Grb2 is made of an SH2 domain able to bind to tyrosine kinase receptors. Thus, Grb2 is able to bind to the activated form of the Epidermal Growth Factor Receptor (EGFR). EGFR activation mainly comes from the binding of a ligand. There is a wide range of ligands that are able to bind EGFR yet, the majority of the ligands come from the ErbB family. The most known ligands are TGF-β and EGF. The binding of these latest induces EGFR dimerization. This dimerization activates the intracellular tyrosine kinase domain characterized by the autophosphorylation of tyrosines (Y992, Y1045, Y1068, Y1086 and Y1173). The activated form of EGFR then recruits Grb2. Indeed, the SH2 domain of Grb2 (from the 60th to the 152nd amino acid) binds the phosphorylated tyrosines of EGFR (Y1068 & Y1086). This interaction recruits SOS (Son Of Sevenless) via the SH3 domain of Grb2. SOS is a GEF protein activating RAS and therefore in turn the MAPK pathway. [18] Therefore, as this example elegantely demonstrates, Grb2 is an adaptor protein able to conduct a signal between two different proteins via its different domains.
DiseasesPhenotypic changes have been identified in cortical and hippocampal neurons characteristic of AD. It seems that Grb2 is implicated in the stimulation of AD. Indeed, the proteins involved in the transduction of the signal from Grb2 to SOS are altered in AD. These modifications would be at the heart of the transduction of a “derived” signal stimulating AD. [19]
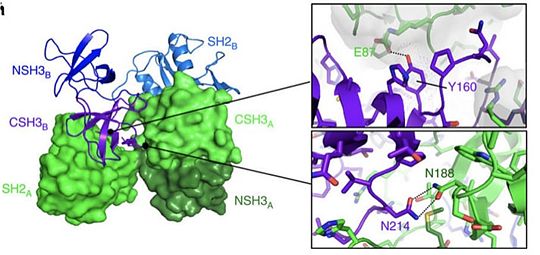
Grb2's isoform could have a simulatory effect in the retro viral infection of HIV-1. By its essential role in the MAPK pathway, Grb3 can have effects on HIV-1 infections. Indeed, the replication of the virus is activated by Lymphocytes T replication. Yet Lymphocytes T’s activation depends on the activation of the MAPK pathway dictated by the presence or not of Grb3 in the cell. This pathway finally activates NFAT, a transcription factor enhancing the LTR promotor of HIV-1 leading to its replication. [20] RelevanceGrb2 protein is especially involved in the setting up of cellular oncognesis in prostate, colon and lung cancers. This role is mainly due to its essential role in signal transduction in the MAP kinase pathway known to induce mitosis. In this pathway, Grb2 binds to the oncogenic protein SOS under its monomeric form. Yet Grb2 can also be found in its dimeric form in the cell. Dimerization of Grb2 is dependent upon several factors like the phosphorylation of or the binding of ligand on the SH2 domain of the same protein. Mainly, phosphorylation induces the dissociation of the Grb2 dimer bringing about an increase in the MAP kinase pathway by the binding of SOS. The phosphorylated state of has been discovered in several pre-metastatis cancers, highly suggesting that pY160 could be a oncogenic marker in humans. A new therapeutic strategy could therefore be considered by stabilizing Grb2 in its dimeric form. This could be achieved with a protein acting as an irreversible cross-link at the interface between the two units. [21]
References
| ||||||||||||