Introduction
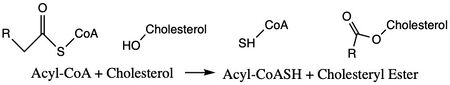
Figure 1. Chemical Structures for the Reactants and Products of ACAT1
Acyl-Coenzyme: A Cholesterol Acyltransferase 1 () also known as Sterol O-Acyltransferase 1 (SOAT1), is an important enzyme in the body.
ACAT1 is an important enzyme that catalyzes the esterification of cholesterol to form cholesterol esters and belongs to the class of enzymes called acyltransferases (Figure 1). As a member of the MBOAT family, it is a key enzyme in lipid metabolism. This enzyme is biologically important because it affects the solubility of cholesterol in the cell membrane and promotes accumulation of cholesterol ester in the cytoplasm as fat droplets. Accumulation of cholesterol ester as these lipid droplets is a main characteristic of
macrophage foaming, which can lead to
atherosclerotic diseases [1].
Cholesterol esters were found in arterial lesions in 1910, but the first ACAT1 activity was discovered in the mid 1900's. This led to the inhibition of ACAT1 as being looked at as a possible strategy of preventing or treating atherosclerosis. Between 1980-1995, the interest in ACAT1 inhibitors grew, but some of the compounds developed exhibited toxicity. As they were looking into the function of the ACAT1 gene, ACAT2 was discovered. In 1993, an ACAT1 gene was successfully cloned. This discovery led to more studies with ACAT1 and atherosclerosis. Some of these studies used mice and also showed cellular toxicity. ACAT1 inhibition is still being pursued as a strategy for treatment or prevention of atherosclerosis and related diseases.
[2]
Structural Overview
ACAT1 was experimentally determined as a of size approximately 260 kDa, composed of helices and loops [3]. Each monomer of the tetramer contains 9 transmembrane helices. However, the was found to be the biologically active arrangement.
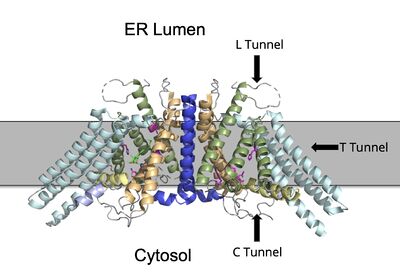
Figure 2. ACAT1 Dimer in the Membrane. The gray shaded region is the plane of the bilipid membrane. The tunnels where molecules enter and exit are labeled. The ER lumen is at the top with the cytosol at the bottom. PBD file: 6P2P.
Dimer-Dimer Interface
The is mobile and mostly hydrophobic, and the residues interact in a shape-complementary manner [3]. It was also found that the reaction chamber is shielded by a lid from the cytosolic side, which leads to low catalytic activity. The binding of Acyl-CoA and cholesterol induce conformational changes that activate the tunnels necessary for substrates to enter them. Work is still being done to fully determine the mechanism of this reaction, but this is the proposed pathway [1].
Tunnels
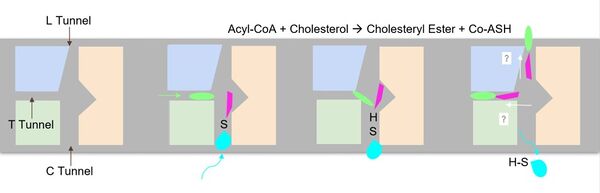
Figure 3. Schematic of the tunnels shown as a cartoon representation
[1].
The active site is accessed through three tunnels that lead from the lumen, cytosol, and transmembrane space to the center of the catalytic site (Figure 3). The tunnels allow the introduction of reactants into the acyltransferase mechanism and the exit of the products to the correct location depending on their function (Figure 2). The cholesterol enters through the T tunnel while the Acyl-CoA enters through the C tunnel. [1]
The is open to the cytosolic side of the protein in which the Acyl-CoA enters into the catalytic domain.
The is the transmembrane tunnel in which the cholesterol enters into the catalytic domain space. Important include Arg262, Phe263, and Leu306. These residues are important for the proper entrance and orientation of the cholesterol to allow for its deprotonation in the mechanism. Upon mutation of these residues, the tunnel function was inhibited. [1]
The provides a potential opening to the lumen side. The enzymatic reaction occurs at the intersection of these two tunnels. It is catalyzed at the intersection of the two tunnels, where the His460 residue is located. The CoASH is released to the cytosol by way of the C tunnel, but the cholesterol ester either exits from the T tunnel to the membrane or through the L tunnel to the lumen. The specific mechanism by which the cholesterol ester product exits the tunnel to enter the lumen has not yet been determined.
Active Site
The active site contains that are essential to the mechanism of the ACAT1 mechanism. These residues include His460 to function as a base catalyst and Asn421 which functions as transition state stabilization with hydrogen bonding. Also important for orientation of the Acyl-CoA ligand is the presence of hydrophobic residues to stabilize the fatty acid (Trp407,Trp420). The active site is at the intersection of all three tunnels to allow a central position for the acyltransferase to occur.
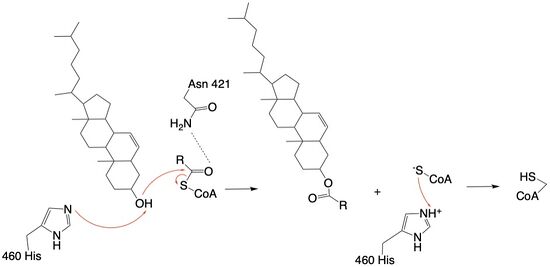
Figure 4: Acyltransferase mechanism of ACAT1 with conserved MBOAT family catalytic residue.
Mechanism
The mechanism of the acyltransferase reaction occurs in the catalytic site of each monomer in the dimer of ACAT1. The T tunnel and and C tunnel converge to the same space to allow the proper orientation of the Acyl CoA and the incoming cholesterol from the transmembrane. The Acyl CoA is oriented in a way to allow the His460 to act as a base catalyst to begin the reaction by deprotonation of the cholesterol which allows it to attack the carbonyl carbon which breaks the sulfur carbonyl bond (Figure 4). The His460 is positioned to deprotonate the cholesterol upon entering through the T tunnel: Acyl-CoA upon entering is positioned to where the sulfur bonded to the carboxyl carbon is at the direct intersection of the T tunnel into the active site. The Acyl CoA is held in place by the of Asn421. This mechanism produces Acyl-CoASH and cholesteryl ester. The Acyl-CoASH leaves through the C tunnel to the cytosol. [1]
Inhibitor
is known as a small molecule inhibitor that is part of the fatty acylamide analog family, and functions as a competitive inhibitor of Acyl-CoA (Figure 5) [3]. Guan discussed that this inhibitor in previous studies had shown that CI-976 reduced the size of atherosclerotic plaques and cholesterol levels in plasma [3] .
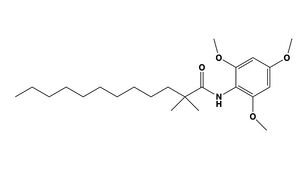
Figure 5. CI-976 Inhibitor
The illustrates how CI-976 can act as a competitive inhibitor of Acyl-CoA. Structurally, Acyl-CoA and CI-976 are both largely hydrophobic, each with long hydrophobic tails. As evident in this image, the hydrophobic tail of CI-976, mimics that of Acyl-CoA. This allows for the CI-976 inhibitor to be recognized by ACAT1 and to bind tightly in the active site pocket, blocking Acyl-CoA from binding, thus rendering ACAT1 unable to perform its reaction.
Diseases
Neurodegenerative Diseases and Cancers
ACAT1 is involved in diseases such as Alzheimer's Disease, Parkinson’s Disease,and other neurodegenerative diseases due to the accumulation of Amyloid Beta (Aβ) plaques in the brain. After research on glioma, prostate cancer, pancreatic cancer, leukemia, and breast cancer, it has been noted that ACAT1 plays a role in the progression of cancer over time. Recently, it was discovered that there was a significant increase in ACAT1 expression in ovarian cancer cell lines [4]. ACAT2 is believed to be upregulated in Nephrotic Syndrome which can lead to cardiovascular disease and renal diseases [5]. Because of ACAT1's activity in tissues such as the aorta, intestine, and liver, it plays a role in atherosclerosis. Studies have shown that the inhibition of ACAT2 can slow the progression of atherosclerosis [6]. Guan discussed a previous study which found that CI-976 decreased the size of atherosclerosis plaques and the overall concentration of cholesterol in the blood plasma of animals that had been fed a high cholesterol diet [3].
Alzheimer's Disease
Alzheimer's Disease is a neurodegenerative disease characterized by accumulation of extracellular plaques that cause interferences with memory retrieval. These plaques are made up of Amyloid Beta (Aβ) peptides which are products of the cleavage of Human Amyloid-Beta Precursor Protein (hAPP) [7] [8]. Within the cells, there is an accumulation of hyperphosphorylated Tau protein. Research has shown that the concentration of cholesterol within cells can affect the production of Aβ [7]. As the concentration of cholesterol in the endoplasmic reticulum of neurons increases, hAPP is downregulated [7]. Inhibition of ACAT1 would lead to higher concentrations of cholesterol in the cells, signaling downregulation of hAPP. Less hAPP available decreases the amount of Aβ peptides being produced which then reduces the available Aβ peptides that form the extracellular plaques associated with Alzheimer's Disease [7] [8].





