Introduction
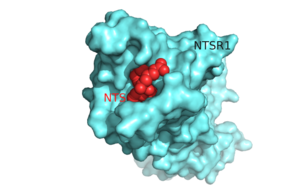
Figure 1.Top view of NTSR1 protein (blue) interacting with its ligand, NTS(red).
Neurotensin receptor 1 (NTSR1) is a G-protein coupled receptor (GPCR). GPCRs are a class of proteins with an extracellular binding domain and 7 transmembrane helices that assist in propagating a cellular response[1]. This is accomplished by the binding of ligands to the GPCR outside the cell, causing a conformational change and activating a signal transduction pathway via second messengers such as cyclic AMP, inositol triphosphate, and diacylglycerol.[1] The ligand for NTSR1 is the 13 amino acid peptide, neurotensin (NTS)[2], and the majority of the effects of NTS are mediated through NTSR1[2]. NTS has a variety of biological activities including a role in the leptin signaling pathways [3], tumor growth [4], and dopamine regulation [5]. Recently NTSR1 was crystallized bound with the C-terminus of its tridecapeptide ligand, . The shortened ligand was used because it has a higher potency and efficacy than its full-length counterpart[2]. Class A GPCRs bind their ligands within the transmembrane core in a ligand binding pocket. The in NTSR1 is located at the top of the protein (Figure 1). NTSR1 also contains an allosteric , which is located directly beneath the ligand binding pocket and the two pockets are separated by the residue [1]. NTSR1 has been mutated to exist in both and states. This has led to a greater understanding of the structure of NTSR1 and how the structure influences its function.
Structure
Ligand Binding Pocket
On the extracellular side of the protein is the
. [2]
One key residue in this pocket is a Phenylalanine at position 358, which takes part in a network of hydrophobic stacking interactions[1]. These interactions stabilize the Trp321 and Tyr324 residues allowing Tyr324 to interact with the C-terminal
via Van der Waals interactions .[2][1]
Without the hydrophobic stacking interactions that are facilitated by the Phe358, this binding interaction would be destabilized. Trp321 also participates in these stacking interactions and serves as the boundary between the ligand binding pocket and the Na+ binding pocket.[1]
Na+ Binding Pocket
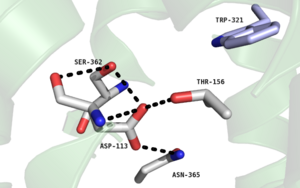
Figure 2. Residues of the collapsed sodium binding pocket. Trp321 (blue) sets the top of the pocket, where Ser362, Asp113, Thr156 and Asn365 (gray) are involved in hydrogen bonding interactions preventing the coordination of a Na
+ ion.
, which is positioned at the bottom of the , sets the top of the . The Na+ ion binding pocket acts as a negative allosteric site for G protein activity [1]. When Na+ enters the Na+ ion binding pocket, it coordinates with Asp95, Gln131, Ser135, and Asp113, decreasing the signaling activity of NTSR1 [1]. When NTSR1 is in its active state, the Na+ ion binding pocket is collapsed. This prevents the regulation of protein activity through a Na+ ion, as the Na+ ion is unable to coordinate via a salt bridge to Asp113 (Figure 2). The side chain atoms of Asp113 form a hydrogen bond network with Thr156, Ser361, Ser362, and Gln365 instead, which prevents the coordination of a Na+ ion[1] (Figure 2).
Activation of NTSR1
Since wild type NTSR1 was unstable in detergent solution for imaging, six residues in the protein were mutated for stabilization.[2] [1]
Active-Like State
The six amino acid mutations for thermostabilization [2] were Ala86Leu, Glu166Ala, Gly215Ala, Leu310Ala, Phe358Ala, and Val360Ala. This protein was found to have NTS affinity similar to that of wild tpye NTSR1, and was named . Along with this, the Na+ ion binding pocket was collapsed in this protein. However, NTSR1-GW5 did not have G-protein activity [2].
Active State
After determining the original structural state, , as only active-like, the structure of NTSR1 was determined in an active state.[1] By reverting back three of the original six mutations from the active-like structure on the basis of their location[1], NTSR1 gained near wild-type activity.[1] The three reversions were Asp166, Leu310, and Phe358, and this protein was named . The revival of activity in NTSR1 indicated that the reverted amino acid residues (Asp166, Leu310, and Phe358) play significant roles in G-protein activity.[1]
Leu310
is crucial for interactions with the G alpha subunit by positioning Arg167 in the conserved [1]. When Leu310 was substituted with alanine, Arg167 was able to form a stabilizing hydrogen bonding network with Asn257, Ser164 and Gly306, which oriented Arg167 in a position that was unfavorable for contacting the G alpha subunit. When residue 310 was converted back to leucine, this hydrogen bonding network was sterically unfavorable and Arg167 interacted with the G alpha subunit[1] leading to the transduction of several different signals involved in dopamine regulation[5], leptin signlaing[3], and tumor growth[4].
Phe358
When this residue was mutated to an alanine [1] the of the ligand binding pocket were interrupted,resulting in a lack of G-Protein activity in NTSR1.[1].This supported the role of Phe358 as stated in the hydrophobic binding pocket section of this page.
Glu166
Although the role of in G-protein activity is not quite as clear as it is for or , substituting this residue for an alanine significantly reduced catalytic G-protein activity [1]. Glu166 is part of a that is highly conserved in class A GPCRs and includes Arg167 and Tyr168. It's hypothesized [1] that Glu166 interacts with Val102, Thr101,and His105 to stabilize the G protein. An important connection between the D/ERY motif and intracellular loop 2 via M181, has also been hypothesized.[1]. ICL2 plays a role in the dissociation of the receptor-G protein complex with GTP present.[1]
Biological Relevance
Neurotensin
is a 13 amino acid peptide that is found in both nervous and peripheral tissues [1]. It functions as a hormone and a neurotransmitter by activating the G-protein coupled receptor, NTSR1[1].
Leptin Research
NTSR1 deficient mice were not able to receive a satiety signal from Leptin[3]. The mice continued to eat when food was present, leading to significant weight gain. With an NTSR1 deficiency, NTS does not bind efficiently to NTSR1, and the leptin signaling pathway is interrupted [3].
Cancer Studies
Some tumor cells can secrete and express NTS and NTS receptors themselves suggesting that NTS autocrine, endocrine and paracrine regulation are possible. This leads to aggressive growth and tumor development. Injecting animals with NTS increased tumor growth and size, while injecting them with NTS antagonist had the opposite effect [4]. NTS regulation may be used in future cancer treatments.
Dopamine Regulation
The dopamine hypothesis states that hyperdopamine levels may lead to schizophrenic symptoms. NTSR1 causes a blockade which inhibits firing in dopaminergic cells suggesting that NTSR1 could be used in schizophrenia treatment. However, this led to extreme secondary effects and was discontinued. Despite this, research on NTSR1 as a treatment for schizophrenia persists[5].


