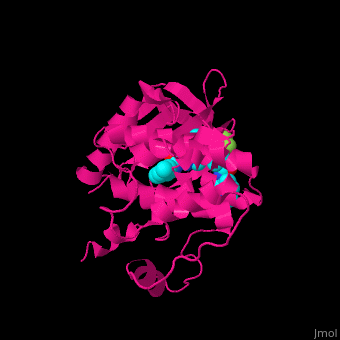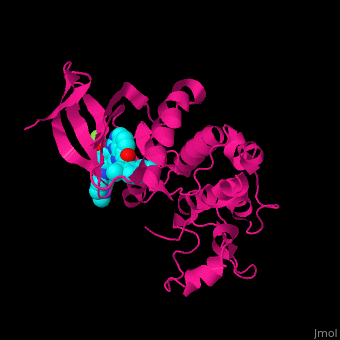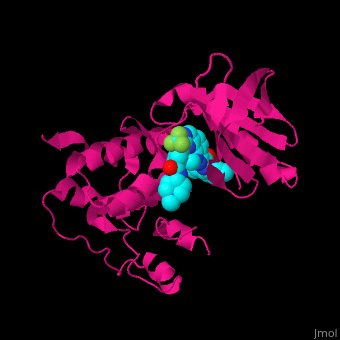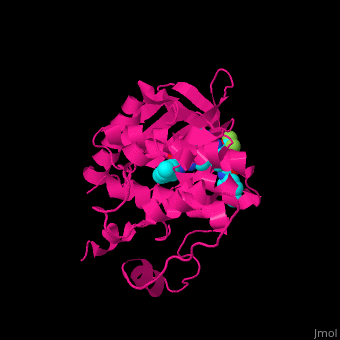Function
Dual specificity protein kinases are kinases which act as both tyrosine kinase and serine/threonine kinase.
- CLK1 phosphorylates serine/arginine-rich proteins involved in pre-mRNA processing.
- Clk2 and Clk3 cause the redistribution of SR proteins and can regulate the alternative splicing of precursor mRNA[1].
- CLK4 phosphorylates tyrosine or serine/threonine[2].
Structural highlights
The dual specificity protein kinase TTK or monopolar spindle kinase 1 contains tetratricopeptide repeat domain (TPR) which serves as interaction domain between proteins. The TPR domain is a helical 34 amino acid motif which is found in several copies in a protein which fold together forming a protein-protein binding domain. Cancer therapy potential drugs bind in .[3][4]
3D structures of dual specificity protein kinase
Dual specificity protein kinase 3D structures