Introduction to ModG
ModG is a cytoplasmic molybdate-binding protein exclusive to the aerobic nitrogen-fixer Azobacter vinelandii.[1] Molybdate is a molybdenum oxyanion (MoO42-). The group 6 element molybdenum is required by many enzymes that catalyze reactions associated with carbon, nitrogen, or sulfur metabolism.[2] It is also part of the cofactor of the molybdoenzyme ModG. Not surprisingly, studies have linked ModG to molybdenum homeostasis within the cell.[1]
Structure
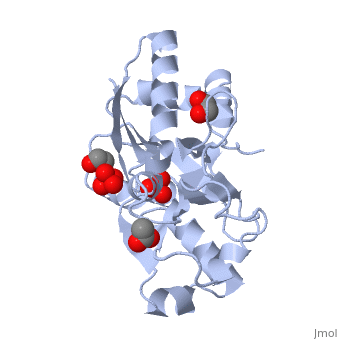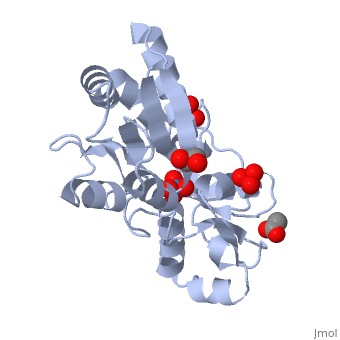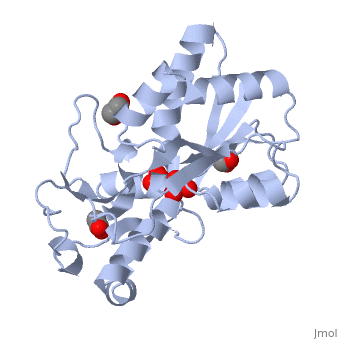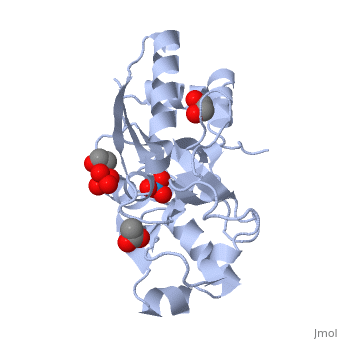
The molybdoenzyme is a homotrimer.[1] It can bind up to 8 molybdate molecules between 4 different types of on subunit interfaces (BS1, BS1’, BS2, and BS2’). Binding site 1 and binding site 2 are found at opposite ends of the protein; binding site 1’ and binding site 2’ are found off-axis near like ends.[3] The sites are connected by hydrogen bonds and thus a cooperative binding mechanism has been proposed for ModG whereby ligand engagement with type 2 sites induces conformational changes to asparagine residues at type 1 sites, reading the site for ligand interactions.[4] The structure of ModG was solved by Delarbe et al. using multi-wavelength anomalous dispersion (MAD).[5] Crystallization required salt-free conditions established with polyethylene glycol (PEG), at which point the authors solved the PEG crystal form using molecular replacement.[5]
The protein is made up of 3 identical subunits that have 67 amino acid pairs and are 14.3 kDa in size.[1] In trimer form the subunits are perpendicular to each other and intersect at the 3-fold axis (as seen in the crystallized form).[3] Each subunit is composed of two β-barrel domains (Domain I and Domain II) that each feature a short 310-helix.[3] Each β-barrel domain includes five antiparallel β-strands arranged in a motif that is capped by two-turn α-helices.[3] The folding arrangement of the ModG domains is that of an oligomer-binding (OB) fold, characteristic of toxins and other intracellular oxyanion-binding proteins.[3] In trimerization, N and C termini (both found in Domain I) of a subunit interact with the β5-β6 loop (Domain II) of the adjoining subunit by sharing antiparallel β-sheets. Approximately 40% of monomer surface area is buried when the protein is in trimer form.[1]
Properties and binding affinity
About 60% of each subunit interface is (40% is polar); furthermore, there is an unequal charge distribution that is attributed to the presence of 4 side chains.[2][4] These include: Lys60, Lys132, Arg6, and Arg78.[1] The are both found on the type 2 binding sites and are entirely buried in the protein; the are partly exposed.[1] This leaves the interface particularly electropositive; electrostatic repulsion is balanced by several favorable interactions that include the formation of 24 hydrogen bonds, 2 salt bridges and approximately 24% of total hydrophobic surface buried (per subunit).[4] The trimer is further stabilized by the binding of an oxyanion which would have a neutralizing effect.[3]
Type 1 binding sites utilize hydrogen bonds and interactions with uncharged polar residues to bind molybdate.[4] The tight pocket volume and rigidity of the site suggest that there is high selectivity for molybdate (or tungstate because ModG cannot differentiate between the two oxyanions).[4] Type 2 sites are comparatively larger in pocket volume, supple, and feature electropositive lysine residues that would contribute to high affinity for negatively charged oxyanions like molybdate.[4] Because the type 2 sites are larger and more flexible, there would be less specificity for molybdate and thus other oxyanions such as phosphate would likely compete for binding.[3][4]
Function in Bacteria
The ability of A. vinelandii to differentiate between molybdate and other oxyanions is of particular interest to researchers.[1][2][5] Phosphate is distinguished from molybdate by the divergence in protonation states: phosphate is protonated at physiological pH whereas molybdate is not.[2] The distinction between sulfate and molybdate is reliant upon differences in ligand size.[2] Interestingly, molybdate permeases are not able to distinguish between tungstate and molybdate.[1][2] Regulation of these intracellular metabolites is attributed to other molybdate-binding proteins collectively known as molbindins.[1][5]
Fate
ModG is eventually degraded and incorporated into molybdopterin, a cofactor of molybdenum enzymes, or the iron-molybdenum cofactor of a nitrogenase enzyme.[1][2] Currently there is little known about cofactor biosynthesis involving the ModG protein.[1]