User:Jamie Abbott/Sandbox1
From Proteopedia
Contents |
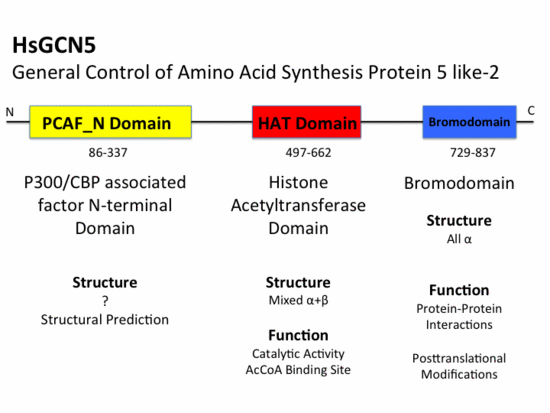
Histone Acetyltransferase GCN5
Histone Acetyltransferase (HAT) GCN5 is a 94 kD protein nuclear HAT or A-type HAT composed of 837 amino acids. GCN5 belongs to the GCN5-related N-acetyltransferase (GNAT) superfamily that includes HATs, aminoglycoside N-acetyltransferases, mycothiol synthase, protein N-myristoyltransferase, and the Fem family of amino acyl transferases.[1] Most HATs function in vivo as members of large multisubunit complexes, many of which were initially characterized as transcriptional regulators. GCN5 has been shown to be part of the STAGA (SPT3-TAFII31-GCN5-L acetylase)[2] complex as well as the TFTC (TATA-binding protein-free TAFII containing)[3] complex. GCN5 is involved in transcriptional regulation by catalyzing the reversible acetylation of lysine residues at position 8 and 16 of H4 and 14 of H3 in vivtro [4]. Acetylation results in the neutralization of charged lysine residues which is hypothesized to weaken histone:DNA contacts[5] as well as alter histone:histone interactions[6]. Chromatin modification more, specifically reversible histone acetylation, has been associated with gene activation and consequently transcriptional activity for many years.
Function and Catalysis
GCN5 catalyzes the transfer of an acetyl group from acetyl coenzyme A () onto the ε-amino group of specific lysine residues presenton N-terminal tails of each of the core histones, H3 and H4, resulting in the neutralization of a single positive charge. [7] Currently, it has been demonstrated that the catalytic mechanism for yeast GCN5 involves glu173 that acts as a general base. The Glu173 residue must deprotonate the ε-amino group of Lys14 of histone H3 prior to attack on the carbonyl carbon of AcCoA. [8] Comparing the sequence of human GCN5 HAT domain with yeast GCN5 strongly suggests that the catalytic mechanism of acetylation would be very similar. The conserved glutamic acid 173 of yeast GCN5 aligns with glutamic acid 575 of human GCN5 and therefore most likely functions as the general base in acetylation of histones H3 and H4.
|
HAT Domain
The HAT domain of human GCN5 [9] consists of amino acid residues 496-658 with mixed α+β secondary structure. The superfamily classification of SCOP places the HAT domain in the N-acetyl transferase (NAT) superfamily. The CATH classification of HAT domain specifies that this α/β protein structure is part of the 3-layer (αβα) sandwich class. This mixed α+β structure consists of six and seven anti-parallel . Structural elements that are a part of the (αβα) sandwich architecture include a and a . The mixed α+β structure of the HAT domain forms an surrounded by helices on both sides. The first four β sheets (β1,β2,β3,β4) and the last three β sheets (β5,β6,β7) of the HAT domain pack against each other in an anti-parallel fashion. The β-hairpin loop motif is stabilized by the between the backbone of β3 and β4. However, β4 and β5 pack against each other forming a parallel . This parallel β-sheet creates a wide cleft in the HAT domain structure. While residues involved in hydrogen bonding to AcCoA largely reside in the loop and α-helix that connects these two sheets; Glu575 believed to be the active site residue, is located in β3 sheet. AcCoA sits on top of this and is to residues in both β3 and β4 sheets as well as the connecting helix. Residues involved in hydrogen bonding AcCoA include Val587, Gly589, Gly591, Thr592, Cys579, and Tyr617. There are two long flexible loops in the HAT domain. links helix α1 and strand β1. connects helix α7 and strand β7 and has been associated with substrate binding and specificity. [10] [11]
Weak Stabilizing Interactions I
Cation-Pi Interactions are weak stabilizing interactions that contribute to the integrity of protein structures. A Cation Pi Interaction involves the coordination of a positively charged residue to the face of an aromatic side chain. Often this electrostatic interaction often involves charged Arg and Lys residues coordinated to aromatic side chain of Phe and Trp residues. The aromatic nature and pi orbitals of a Trp or Phe residue create a partial negative charge at the center of the face and assist in coordination of the cation side chain of a Arg or Lys residue. The HAT domain contains a interaction that involves residues Trp519 and Arg515.
|
Bromodomain
The bromodomain is a highly conserved domain found to be a part of many chromatin remodeling proteins such as HATs. The Bromodomain of GCN5 falls into the SCOP all alpha protein class, which corresponds, to the CATH mainly alpha class category with up-down bundle architecture. This 110 amino acid motif was originally identified as a sequence motif common to the Drosophila brahma and female-sterile homeotic proteins, the yeast SWI2/SNF2 proteins and the human CCG1 protein [12][13] While the bromodomain 60 residues away from HAT domain no evidence suggests that it is involved in or necessary for histone acetyl transferase activity.
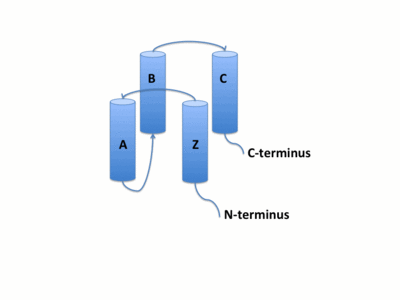
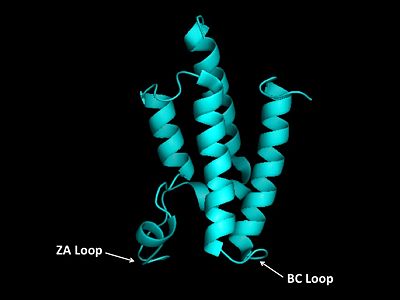
The GCN5 bromodomain is located near the C-terminus and is a 71% α-helical domain of 110 amino acids with no β structures. The bromodoamin forms a consisting of helices αZ,αA,αB,and αC. In this four helix bundle the anti-parallel adjacent α-helices pack together with angles approximately 18° to 20° to the helical axes. The four helical-bundle arrangement commonly forms the "Ridges and Grooves" model of helical packing. Adjacent αB and αC helices pack together forming the model and allows for residues from one helix to fit into the grooves of the other. Additionally, the up-and-down four-helix bundle of the GCN5 bromodomain has left handed topology as a result of the orientation of the long ZA loop.
Weak Stabilizing Interactions II
Salt Bridge Formation is another example of weak interactions that can stabilize the secondary structures of proteins. The formation of salt bridges can increase packing density, increase hydrophobic core packing, and even decrease the length of surface loops.[14] The formation of salt bridges involves both hydrogen bonding and electrostatic interactions and frequently occur between the anionic carboxylate side chain of either Asp of Glu and cationic ammonium side chain of Lys or Arg. Other residues with ionizable side chains, such as His, Tyr, and Ser may also participate in the formation of salt bridges. Salt bridge charge-charge interactions can be characterized in two ways. First, that the cation and anion are hydrogen bonded to each other or second, that the cation and anion are not hydrogen bonded to each other. The distance of salt bridges typically do not exceed 3.5 Å. The bromodomain has five involving residues R805, E822, K767, R770, E813, R816, E828, D831, E780, and K776. One of these bridges involves the stabilization of helices αB and αC while another forms in the AZ Loop and may contribute the stabilization of the 3z helix.
Protein-Protein Interactions
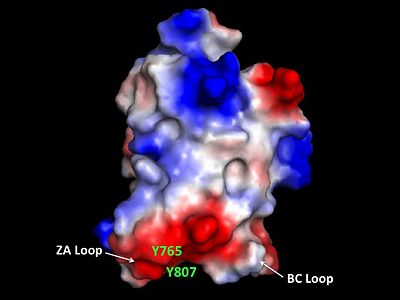
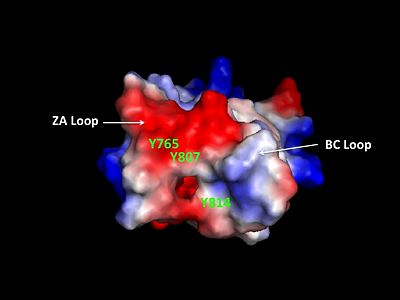
The bromodomain contains two long distinct loops connecting adjacent helices. Helices αZ and αA are connected by the ZA loop while helices αB and αC are connected by the BC loop. A two additional helices exist in the long ZA loop termed 3z and αA'. The ZA and BC loops pack together to form a hydrophobic pocket that may be involved in protein-protein interactions. [15] The is lined with residues Val757, Ala762, Tyr765, Tyr807, and Tyr814. The surface of this pocket is essentially the surface of the hydrophobic core of the bromodomain and some of the same residues that line this hydrophobic pocket are involved in the formation of the hydrophobic core. Residues that line the interior of the include F753, V757, Y765, I773, D774, V800, N803, C804, Y807, N808, and Y814. The bromodomain is separated from the HAT domain by 57 amino acids residues that also contain an ADA2 protein interaction site.[16]
Post-Translational Modifications
GCN5 contains one known site for posttranslational modification. A tyrosine residue at position 734 is subject to phosphorylation by tyrosine kinases. [17] [18] Tyr734 is located in the helix of the bromodomain. The may act as a molecular switch that serves to activate or deactivate GCN5 and more specifically affect the bromodomain.
PCAF_N Domain
Structural Prediction
There is no known structure for the PCAF_N domain or p300/CBP assocaited factor N-terminal domain. From the known sequence for the PCAF_N domain, structural predictions can be generated for this domain. PredictProtein can predicted protein structural information from a sequence; such as secondary structure, solvent accessibility, transmembrane helices, globular regions, coiled-coil regions ,structural switch regions, disorder regions, protein-protein and protein-DNA binding sites, sub-cellular localization, beta-barrels, cysteine bonds, metal binding sites and disulphide bridges.
Using PredictProtin the PCAF_N domain was predicted to be a mixed α+β structure (31% helical, 4% beta, and 65% loop). Other important structural features that were predicted included one disulfide bond and three sites for postranslational modifications. Posttranslational modification sites included a N-glycosylation site, a Casein kinase II phosphorylation site, and a N-myristoylation site. N-linked glycosylation sites most often serve as locations for the addition of sugars co-translationally to aid in protein folding. Myristoylation is the addition of C-14 FA myristate to the N-terminus of a protein. This type of modification can take place either post-translationally or co-translationally. Myristoylation is irreversible and necessary for interaction with the cytosolic membrane.
Also, by taking advantage of Protein Homology/analogY Recognition Engine or PHYRE3D models for the PCAF_N domain can be generated based on the primary sequence.
Evolutionary Conservation
Structural Homology
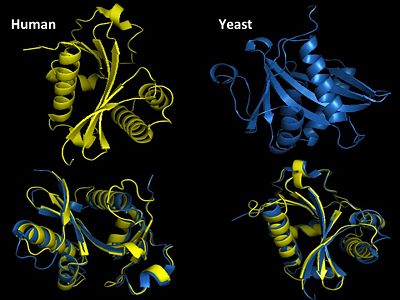
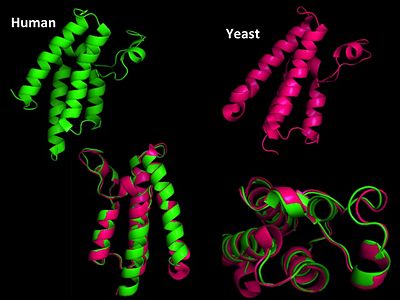
GNAT super family (GCN5-related N-acetyltransferase) family has both both prokaryotic and eukaryotic members sharing identity in four conserved motifs[19]. Motif A is the most highly conserved and is involved in AcCoA recognition and binding and is part of the HAT domain. Yeast GCN5 is one of the best characterized HATs and shares substantial sequence and structural homology to human GCN5. Yeast and human are 98% identical in their sequence also share structural homology in both the HAT and bromodomain. The first HAT catalytic subunit of a A-type HAT was identified in a macronuclear preparation from ciliated protozoan Tertraymena[20]. It was also noted that this enzyme had significant sequence homology with transcriptional regulator yeast GCN5 (yGCN5).
References
- ↑ Vetting MW, S de Carvalho LP, Yu M, Hegde SS, Magnet S, Roderick SL, Blanchard JS. Structure and functions of the GNAT superfamily of acetyltransferases. Arch Biochem Biophys. 2005 Jan 1;433(1):212-26. PMID:15581578 doi:10.1016/j.abb.2004.09.003
- ↑ Martinez E, Kundu TK, Fu J, Roeder RG. A human SPT3-TAFII31-GCN5-L acetylase complex distinct from transcription factor IID. J Biol Chem. 1998 Sep 11;273(37):23781-5. PMID:9726987
- ↑ Brand M, Yamamoto K, Staub A, Tora L. Identification of TATA-binding protein-free TAFII-containing complex subunits suggests a role in nucleosome acetylation and signal transduction. J Biol Chem. 1999 Jun 25;274(26):18285-9. PMID:10373431
- ↑ . PMID:5339769
- ↑ . PMID:5339769
- ↑ Wolffe AP, Hayes JJ. Chromatin disruption and modification. Nucleic Acids Res. 1999 Feb 1;27(3):711-20. PMID:9889264
- ↑ Schuetz A, Bernstein G, Dong A, Antoshenko T, Wu H, Loppnau P, Bochkarev A, Plotnikov AN. Crystal structure of a binary complex between human GCN5 histone acetyltransferase domain and acetyl coenzyme A. Proteins. 2007 Jul 1;68(1):403-7. PMID:17410582 doi:10.1002/prot.21407
- ↑ Tanner KG, Langer MR, Kim Y, Denu JM. Kinetic mechanism of the histone acetyltransferase GCN5 from yeast. J Biol Chem. 2000 Jul 21;275(29):22048-55. PMID:10811654 doi:10.1074/jbc.M002893200
- ↑ Schuetz A, Bernstein G, Dong A, Antoshenko T, Wu H, Loppnau P, Bochkarev A, Plotnikov AN. Crystal structure of a binary complex between human GCN5 histone acetyltransferase domain and acetyl coenzyme A. Proteins. 2007 Jul 1;68(1):403-7. PMID:17410582 doi:10.1002/prot.21407
- ↑ Clements A, Poux AN, Lo WS, Pillus L, Berger SL, Marmorstein R. Structural basis for histone and phosphohistone binding by the GCN5 histone acetyltransferase. Mol Cell. 2003 Aug;12(2):461-73. PMID:14536085
- ↑ Poux AN, Marmorstein R. Molecular basis for Gcn5/PCAF histone acetyltransferase selectivity for histone and nonhistone substrates. Biochemistry. 2003 Dec 16;42(49):14366-74. PMID:14661947 doi:10.1021/bi035632n
- ↑ Owen DJ, Ornaghi P, Yang JC, Lowe N, Evans PR, Ballario P, Neuhaus D, Filetici P, Travers AA. The structural basis for the recognition of acetylated histone H4 by the bromodomain of histone acetyltransferase gcn5p. EMBO J. 2000 Nov 15;19(22):6141-9. PMID:11080160 doi:10.1093/emboj/19.22.6141
- ↑ Tamkun JW, Deuring R, Scott MP, Kissinger M, Pattatucci AM, Kaufman TC, Kennison JA. brahma: a regulator of Drosophila homeotic genes structurally related to the yeast transcriptional activator SNF2/SWI2. Cell. 1992 Feb 7;68(3):561-72. PMID:1346755
- ↑ Dhalluin C, Carlson JE, Zeng L, He C, Aggarwal AK, Zhou MM. Structure and ligand of a histone acetyltransferase bromodomain. Nature. 1999 Jun 3;399(6735):491-6. PMID:10365964 doi:10.1038/20974
- ↑ Goldman A. How to make my blood boil. Structure. 1995 Dec 15;3(12):1277-9. PMID:8747452
- ↑ Candau R, Zhou JX, Allis CD, Berger SL. Histone acetyltransferase activity and interaction with ADA2 are critical for GCN5 function in vivo. EMBO J. 1997 Feb 3;16(3):555-65. PMID:9034338 doi:10.1093/emboj/16.3.555
- ↑ Rush J, Moritz A, Lee KA, Guo A, Goss VL, Spek EJ, Zhang H, Zha XM, Polakiewicz RD, Comb MJ. Immunoaffinity profiling of tyrosine phosphorylation in cancer cells. Nat Biotechnol. 2005 Jan;23(1):94-101. Epub 2004 Dec 12. PMID:15592455 doi:nbt1046
- ↑ Rikova K, Guo A, Zeng Q, Possemato A, Yu J, Haack H, Nardone J, Lee K, Reeves C, Li Y, Hu Y, Tan Z, Stokes M, Sullivan L, Mitchell J, Wetzel R, Macneill J, Ren JM, Yuan J, Bakalarski CE, Villen J, Kornhauser JM, Smith B, Li D, Zhou X, Gygi SP, Gu TL, Polakiewicz RD, Rush J, Comb MJ. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell. 2007 Dec 14;131(6):1190-203. PMID:18083107 doi:S0092-8674(07)01522-X
- ↑ Roth SY, Denu JM, Allis CD. Histone acetyltransferases. Annu Rev Biochem. 2001;70:81-120. PMID:11395403 doi:10.1146/annurev.biochem.70.1.81
- ↑ Rush J, Moritz A, Lee KA, Guo A, Goss VL, Spek EJ, Zhang H, Zha XM, Polakiewicz RD, Comb MJ. Immunoaffinity profiling of tyrosine phosphorylation in cancer cells. Nat Biotechnol. 2005 Jan;23(1):94-101. Epub 2004 Dec 12. PMID:15592455 doi:nbt1046