You may include any references to papers as in: the use of JSmol in Proteopedia [1] or to the article describing Jmol [2] to the rescue.
Introduction
Beginning in 2000, the National Institutes of Health’s Protein Structure Initiative was a fifteen-year program that worked to solve the structures of many proteins with known DNA sequences. The present study analyzes one such protein, known as , a predicted alpha/beta hydrolase found in Escherichia Coli. The specific objective of this study is to functionally characterize 3H04 based on structural analysis of the protein. In silico enzyme characterization for 3H04 was carried out using BLAST, Pfam, and Dali servers, followed by active site analysis using the PyMOL plug-in ProMOL. This structural and protein sequence analysis suggests PDB entries and , respectively, are most similar to 3H04; however 1TAH and 3H04 share the most catalytic triad similarity. To investigate the lipase activity of 3H04 in vitro, recombinant his-tagged protein was isolated from E. coli.
Proteins are macromolecules that are present in nearly every earthly organism and represent a vast array of functions. Scientists have been able to structurally solve proteins at a faster rate than they have been able to identify the functions of these same proteins. In an effort to close this gap, the National Institutes of Health launched the Protein Structure Initiative, which was a fifteen-year program, starting in 2000, that worked to solve the structures of many proteins with known DNA sequences. However, there are still hundreds proteins housed within the Protein Data Bank that have unidentified functions. Researchers are continuing to advocate for further analysis of these proteins through the development of BASIL modules that explain how to use ProMOL to analyzes the structural similarities between the active sites of proteins.
Overview
This project seeks to determine the function of a protein with an identified structure. The query protein, in this case, can be identified by the Protein Data Bank Identification (PDB ID) code . 3H04 is a alpha/beta hydrolase found in Escherichia Coli, a bacterium that resides in the lower intestines of warm-blooded mammals. The more specific purpose of this study is to characterize the function of 3H04. Plasmid purification is just one method for identifying an unidentified protein. Protein purification in bacterial cells requires the plasmids, a small circular double-stranded DNA molecule, to be purified and identified. The purification of the plasmid is meant to isolate and purify the plasmid DNA. The protein in the plasmid is identified through bacterial cells, and grows against ampicillin so the bacterial cells are growing against the antibiotic; the cells that grow have AmpR (Ampicillin Resistance gene). And in efforts to characterize our protein, 3H04, we are comparing it to other proteins with significant parts in common to hopefully help characterize the function of 3H04. The comparison of the different aspects of our protein to different aspects of other proteins also helped us determine our assay.
Hypothesis
In the beginning, we predicted that our protein was an esterase, because originally our most promising homologs were an aminopeptidase and an esterase. And even though the most hits from our Promol search were enzyme class 3.4 (aminopeptidases), no further information was found about the catalytic triads of these hits. We decided to use a homolog in E.C. 3.1 (esterase) because there was information about their catalytic triads. We knew that our protein is a hydrolase, and we believe that 3H04 is characterized as an esterase (3.1), instead of an aminopeptidase (3.4). Even though the 3.4 category proteins had the most motif structure similarity in Promol, we chose (a lipase) as our target homolog. 1TAH is a characterized protein and has a similar catalytic triad to what we predict for our protein (3H04). When we aligned the two catalytic triads and , they had an RMS of 0.016. In the beginning we were looking for a homolog with more protein sequence and structure similarity, but because neither protein sequence or structure determine the function of 3H04, we decided that those two factors matter much less than the catalytic triad of the protein. So, we used Promol to find what the catalytic triad of 3H04 is and we used 1TAH as our target homolog for the catalytic triad. We predict, because the RMS was so low, that 3H04 and 1TAH will be having very similar enzymatic functions.
Methods
We performed a Lipase Assay to determine what kind of esterase our protein is, because our target homolog (1TAH) is a lipase. The literature about 1TAH had no useful information about assays, but we used an assay from a paper regarding a different lipase. We knew that our protein was a hydrolase, and we believed it to be an esterase, and with this lipase assay we were able to deduce that our protein was an esterase and a lipase. To determine if 3H04 functions as a lipase on the p-Nitrophenyl Butyrate (pNPB), a colorimetric assay using a spectrophotometry protocol. After mixing pNPB in 2-propanol, add 50 mM Tris-HCl buffer (pH 8.0, containing 0.4% w/v Triton XI00 and 0.1% w/v arabic gum) to the mixture. To initiate reaction, add pNPB solution and measure absorbance using a UV spectrophotometer at 410 nm for 5-10 min, recording absorbance values at 20 second intervals.
Results
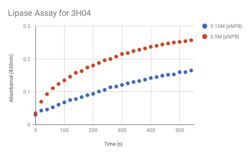
Figure 1: Lipase Assay Result. 3H04 was tested with two different concentrations of pNPB to test how the concentration altered the rate of the reaction.

Figure 2: Coomassie Gel. Coomassie Gel of 3H04 (Lanes 1-8). 1. Cell Extract, 2. Flow Through, 3. Wash 1, 4. Wash 3, 5. Elution 1, 6. Elution 2, 7. Elution 3, 8. Ladder. The protein is being expressed wherever there is a darker, thinker band in the same row as the 75 kDa.
Conclusion
We've concluded that our protein is a lipase, and because we expressed our protein through E.coli, we believe that 3H04 functions in the lower intestine of warm blooded organisms. The blue dots in Figure 1 represent the pNPB at a lower concentration than the red dots. The blue dots represent the reaction happening at 0.15M and the red dots represent the reaction with a concentration of 0.5M, both concentrations showed positive results of a reaction, however the expected curve of the graph was not as steep as predicted with the original concentration of 0.15M. The concentration was increased to increase the curve of the graph so that the expected curve was attained. The stained coomaise gel in Figure 2 shows that our protein was being expressed through the bacterial cells, around the correct expected molecular weight. This protein, expressed, purified, and now characterized is in the enzyme class lipase.


